How To Draw An Atom Step By Step
This twelvemonth, I've been basing my introduction to bones chemistry for my middle school students around the periodic table of the elements. The first step, however, is to teach them how to depict basic models of atoms.
Prep: Memorization over the Winter Break
I started it off by having the students memorize the first 20 elements (H through Ca), in their right lodge — by atomic number — over their wintertime break.
So that they'd take a bit of context, I went over the basic parts of an cantlet (protons, neutrons, and electrons) and made it clear that the proper name of the element is adamant solely by the number of protons. I fifty-fifty had them draw a few atoms with the protons and neutrons in the center and the electrons in shells. Since I'd dumped all of this on them in a unmarried class menses, it probably was a flake much, merely since it was merely to give them some context I did not await the 7th graders, who had not seen this earlier, to think it all; for the 8th graders it should have been simply a review.
Nigh students did a skillful job at the memorization. Some found songs on the the internet that helped, while others just pushed through. Having the two weeks of winter pause to work on it probably helped also.
Day 1. Lesson: The Parts of an Cantlet
When we got dorsum to schoolhouse, the showtime affair I did was give them an outline of the upper part of the periodic table and asked them to fill it in with the element names.

Afterward they'd filled out their periodic tabular array template, I went into the parts of the atoms in more detail, and had them exercise. The primal points I wanted them to remember were:
-
The atomic number is written every bit a subscript to the left of the chemical element symbol. The atomic number is the number of protons. Since they memorized the elements in order, they should be able to figure this out on their ain — but they could also await it up apace on the periodic tabular array, or look at the element symbol where the diminutive number is sometimes written on the lower left.
- The atoms accept the aforementioned number of electrons as protons. Protons are positively charged, and electrons are negatively charged, so an atom needs to have the same number of both for its charge to be balanced. We don't talk about ions –where at that place are more or less electrons– until later.
-
The atomic mass (4) is written equally a superscript to the left of the element symbol. The diminutive mass is the sum of the number of protons (ii) and the number of neutrons (2). The minor atoms that we're looking at tend to have the same number of neutrons every bit protons, but that'south not necessarily the case. So how do you lot know how many neutrons? You lot take to ask, or look at the atomic mass number, which is usually written to the upper left of the atom. Since the atomic mass is the sum of the number of protons and neutrons, if you know the atomic mass and the number of protons, you can hands figure out the number of neutrons. (Note that electrons don't contribute to the mass of the atom considering their masses are and then much smaller than the masses of neutrons and protons.
-
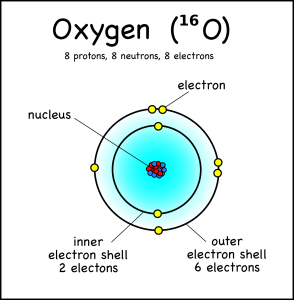
This oxygen cantlet has viii electrons in two shells. Electron Shells: Electrons orbit around the nucleus in a series of shells. Each shell tin hold a sure maximum number of electrons (ii for the first shell; 8 for the second shell; and 8 for the third). And to draw the atoms you fill up the inner shells showtime then movement on to the outer shells.
Then, if I wrote just the element symbol and its atomic mass on the board that students should be able to figure out the number of particles.
Example: Carbon-12
For example, the most common grade (isotope) of carbon-12 is written every bit:
- Protons = 6: Since we know the atomic number is 6 (because we memorized it), the atom has 6 protons.
- Neutrons = 6 : Since the diminutive mass is 12 (upper left of the element symbol), to find the number of neutrons we subtract the number of protons (12 – 6 = 6).
- Electrons = six: This cantlet is balanced in charge and so information technology needs half dozen electrons with their negative charges to outset the six positive charges of the six protons. (Note: we haven't talked almost unbalanced, charged atoms all the same, but the charge will bear witness up every bit a superscript to the right of the symbol.)
- Electron shells (2-4): We have half dozen electrons, so the first two go into filling up the outset electron beat, and the rest tin become into the second vanquish, which can hold upwards to 8 electrons. This gives an electron configuration of 2-four.

Instance: Carbon-14
Carbon-14 is the radioactive isotope of carbon that is often used in carbon dating of historical artifacts. Information technology is written equally:
- Protons = 6: As long as it's carbon it has six protons.
- Electrons = vi: This cantlet is likewise balanced in charge so information technology as well needs six electrons.
- Neutrons = 8 : With an atomic mass of fourteen, when we subtract the six protons, the number of neutrons must exist 8 (14 – vi = eight).
The simply difference between carbon-12 and carbon-14 is that the latter has two more neutrons. These are therefore 2 isotopes of carbon.

Instance: Helium-iv

Case: Sodium-23

Note: A film of a hydrogen atom can be establish hither.
Update: I've created an interactive app that will describe atoms (of the first 20 elements), to become with a worksheet for educatee exercise.
Source: https://montessorimuddle.org/2013/01/13/drawing-atoms/
Posted by: washingtondishemeard.blogspot.com


0 Response to "How To Draw An Atom Step By Step"
Post a Comment